- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
Products
New Products
Pharmaceutical Impurities Manufacturers
Pharmaceutical impurities are active pharmaceutical ingredients (API) or undesirable chemical components in pharmaceutical formulations. Impurities in the bulk drug may originate from the synthesis process or other sources such as raw materials, intermediates, reagents, solvents, catalysts, and reaction by-products. During the drug development process, impurities may be: • formed due to the inherent instability of the API • due to incompatibility with the added excipients, or • interacts with packaging materials and container sealing systems (CCS) to produce APIs The content of various impurities in the final product determines the safety of the final product. Therefore, the identification, quantification, qualitative and control of impurities have become a key component of the drug development process.
- View as
-
-

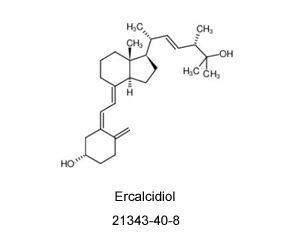
21343-40-8
If you are looking for a supplier or manufacturer of Ercalcidiol (CAS: 21343-40-8), please contact us. For more than 10 years, we have been focusing on custom synthesis and large-scale manufacturing of organic compound products. We can provide high-quality compounds and good service to customers all over the world. Now, we also hope to meet your needs for Ercalcidiol (CAS: 21343-40-8) and other compounds.
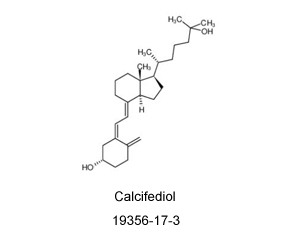
Read MoreSend Inquiry19356-17-3
If you are looking for a supplier or manufacturer of Calcifediol (CAS: 19356-17-3), please contact us. For more than 10 years, we have been focusing on custom synthesis and large-scale manufacturing of organic compound products. We can provide high-quality compounds and good service to customers all over the world. Now, we also hope to meet your needs for Calcifediol (CAS: 19356-17-3) and other compounds.
Read MoreSend Inquiry